Basic Sciences
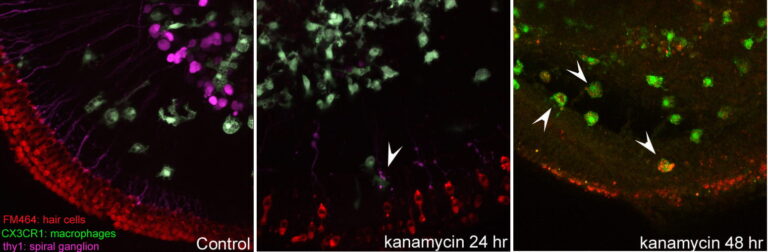
Hirose Lab
After various forms of injury, the inner ear degenerates and inflammatory cells exit the blood vessels and enter the fluid spaces of the cochlea. To what degree these inflammatory cells could be helpful for repair or could serve to exacerbate injury is uncertain. We use methods to image live macrophages in cultures of the mouse inner ear as well as methods to suppress monocytes and macrophages in living mice to determine how these cells affect injury in the cochlea after exposure to ototoxic medications, noise damage or other insults that affect the survival of inner ear sensory cells.
Kim Lab
The ability to obtain multicellular functional images in vivo has transformed neuroscience research, especially the field of sensory physiology. Our novel surgical methods allow real-time visualization of multiple cochlear cells in hearing-preserved animals. We use these approaches to help characterize the properties of sound coding and processing in the cochlea and along the central auditory pathway. Our efforts will also shed light on the transport of ototoxic drugs into the cochlea and promote the development of pathway blockers to prevent drug-induced hearing loss.

Peng Lab
Our mission is to understand the role of the immune system in the pathogenesis of cancer and age-related neurological disorders, and then to develop effective strategies for the manipulation of immune cell fate and function to provide disease prevention and immunotherapy.

Puram Lab
We are a translational/basic science laboratory devoted to understanding the transcriptional and epigenetic heterogeneity of head and neck cancers. By understanding the mechanisms underlying the growth, development and spread of these cancers, we hope to develop new
diagnostics and therapeutics to treat this devastating disease.
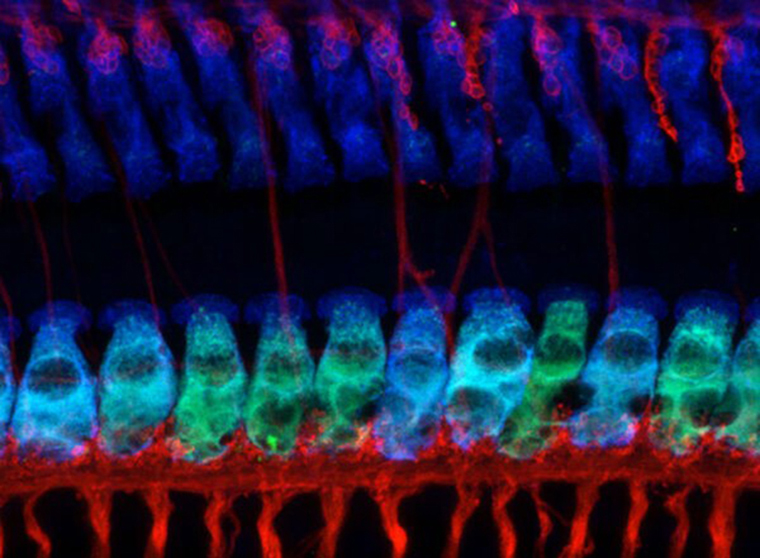
Rutherford Lab
Sensory encoding and excitotoxicity
The Rutherford Lab is focused on sensory encoding. We study how sound is transduced by the inner ear into action potentials in the auditory nerve through mechanisms of synaptic transmission. During overexposure to sound, excessive release of glutamate from the sensory receptors (inner hair cells) drives an excito-toxic response resulting in synaptic disintegration and auditory nerve degeneration. Our experiments are testing hypotheses about the mechanisms of glutamatergic excitotoxicity in the cochlea.
Sensory Regeneration Lab
Sensory transduction in the inner ear is mediated by mechanoreceptive hair cells. Unfortunately, hair cells can be injured or lost after exposure to noise, ototoxic drugs, or as part of normal aging. Such injury typically results in permanent hearing loss and/or disequilibrium. Our research focuses on the biological mechanisms that regulate the production of hair cells and the survival and growth of their afferent neurons.

Sheets Lab
Our laboratory's goals are to understand how specific pathological changes occur in hair cells exposed to noise by defining the dynamic cellular processes that lead to hair-cell synapse loss and hair-cell death. We investigate these questions using zebrafish as a model for human hearing and deafness.

Yakusheva Lab
The goal of our research is to study the role of the vestibulo-cerebellum in motor control, balance and spatial navigation. The vestibulo-cerebellum is a region of the cerebellar cortex that comprises the floccular lobe and the posterior vermis. Currently, our lab is testing the hypothesis that the floccular lobe computes a forward model (predictive model) of the eye movement from vestibular and efferent copy information, and that the posterior vermis computes an estimate of our heading (translation) and orientation in space from semicircular canals and otolith information. Our experimental techniques include single unit recordings, pharmacology, behavioral neuroscience, mouse neurogenetics, histochemistry and computational modeling.